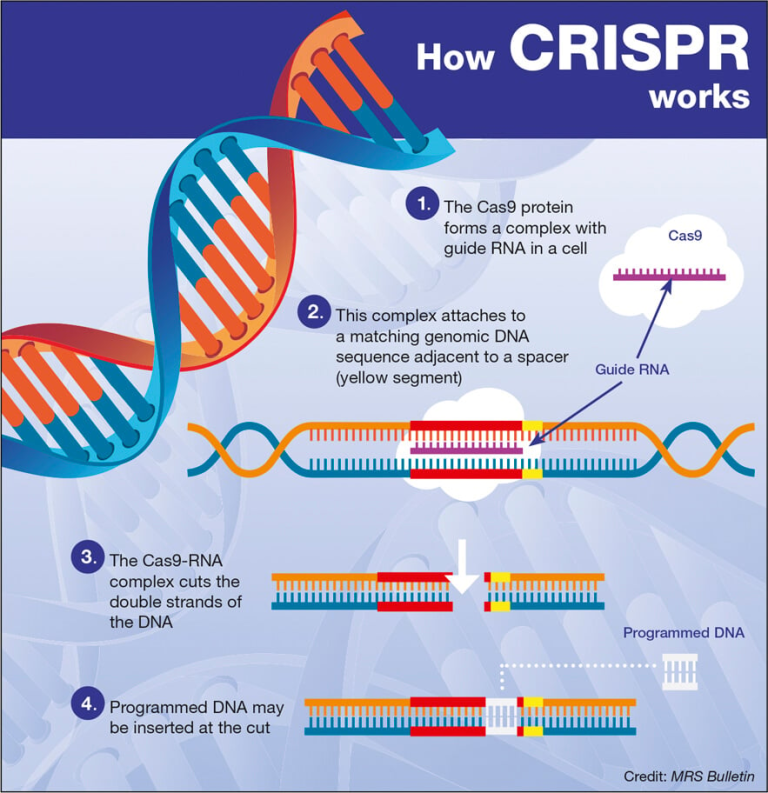
The U.S. health innovation system stands as a beacon of progress and collaboration, often looked upon with admiration by nations around the globe. With its roots tracing back to pivotal moments in history, such as the mass production of penicillin during World War II, this system has evolved into a robust network of public-private partnerships that underpin biomedical research. Federal funding has been a crucial element, enabling scientific breakthroughs that have transformed healthcare and medical technology. Today, the synergy between government entities, academic institutions, and the private sector continues to drive advancements, ensuring that the U.S. remains at the forefront of health innovation. This ongoing journey not only nourishes the nation’s public health landscape but also fuels economic growth, proving that the collaboration of diverse entities can yield extraordinary results.
The framework of health innovation in the United States is characterized by a dynamic interplay of research, technology, and funding. The landscape is often described as an ecosystem of health breakthroughs, deeply influenced by historical achievements and the collaborative efforts of various stakeholders. Specifically, the relationship between federal agencies and private enterprises has fostered significant advancements in biomedical science, particularly in the wake of urgent needs that shaped the nation’s response during major conflicts. This innovative system, which has thrived for decades, continuously integrates new scientific knowledge and technologies, ensuring that the U.S. not only meets its current health challenges but is also prepared for the future. As this complex network of researchers, economists, and policymakers endeavors to sustain its remarkable achievements, the implications for global health and innovation loom large.
The Legacy of Penicillin in U.S. Health Innovation
The story of penicillin is a pivotal landmark within the U.S. health innovation ecosystem. Discovered in the 1920s yet only mass-produced during World War II, penicillin exemplifies how urgent scientific needs can drive rapid advancements. With the war presenting unprecedented threats to soldiers and civilians alike, the U.S. government recognized the dire need for a means to combat bacterial infections that were causing higher mortality than battlefield injuries. This led to a concerted effort to streamline the mass production of penicillin, showcasing the effective collaboration between federal funding, university research, and industrial capabilities. As a result, penicillin became one of the first antibiotics to revolutionize medical treatment not only for soldiers but also for the broader population, marking the beginning of a golden age in pharmaceuticals. The lessons learned from this development period still resonate in today’s biomedical research initiatives, which continue to prioritize rapid response to public health crises.
The development of penicillin also influenced the trajectory of federal funding for biomedical research. The successful mass production efforts were not just an isolated victory; they catalyzed the establishment of a robust infrastructure that would support future scientific breakthroughs. This public-private partnership model became a blueprint for the funding and development of new therapies and technologies in the years that followed. Additionally, the narrative surrounding penicillin emphasizes the importance of sustaining investment in biomedical research to ensure continuous innovation in the face of emerging health threats. The federal government’s role in facilitating this success has become a cornerstone of discussions about funding strategies in modern medicine.
Moreover, the efforts that led to the successful development of penicillin laid the groundwork for additional scientific breakthroughs in the postwar period. Researchers developed new methodologies for identifying and cultivating antibiotics, which directly influenced the rise of a new generation of infectious disease research. The significance of interdisciplinary collaboration became evident, as chemists, microbiologists, and pharmacologists began working together to synthesize and analyze potential therapeutics at a previously unimaginable scale. As a consequence, the U.S. health innovation ecosystem evolved, seamlessly integrating academic institutions with industrial partnerships that paved the way for significant advancements in health care, technology, and pharmaceuticals. In the years to come, penicillin’s legacy would inspire further investments into medical R&D, leading to an array of life-saving treatments and significantly improved public health outcomes.
Federal Funding and Its Role in Biomedical Research
Federal funding has long played a critical role in shaping the landscape of biomedical research in the United States. Since World War II, when the Office of Scientific Research and Development (OSRD) emerged to coordinate research efforts, a strong constitutional framework was established to ensure that public funds could support urgent medical advancements. This institutional support has allowed universities and private sectors to innovate collaboratively, yielding breakthroughs that have transformed public health. Among these advances, the establishment and consistent growth of the National Institutes of Health (NIH) stands out, as it has provided vital funding for research projects, fostering partnerships that drive innovation in drug discovery, medical technology, and disease prevention strategies.
Today, however, the ongoing debate surrounding federal funding allocations highlights the delicate balance between maintaining a vibrant innovation system and the demands for budget cuts. Initiatives that might limit reimbursement for research costs could endanger this long-standing support structure. Without adequate federal investment, many essential biomedical research projects could face significant setbacks, potentially stalling advancements that have become the backbone of the U.S. health innovation ecosystem. Public-private partnerships, particularly in critical fields like cancer research, infectious disease treatment, and chronic disease management, rely heavily on consistent funding levels to sustain their efforts and continue generating impactful results.
In addition to facilitating breakthroughs in medicine, federal funding plays an indispensable role in training the next generation of scientists and researchers. The academic programs supported by such funding not only bolster research capacities but also cultivate a workforce prepared to tackle future health challenges. Many of today’s innovative scientists were nurtured within the infrastructure established by federally funded initiatives during the postwar era. Programs aimed at enhancing research excellence empower universities to attract talent and provide critical training resources that are foundational for successful careers in biomedical research and public health. This robust pipeline of skilled professionals results in sustained progress in scientific inquiry while reinforcing the importance of federal investment in fostering innovation.
Public-Private Partnerships: A Catalyst for Innovation
Public-private partnerships have emerged as a linchpin in the U.S. health innovation ecosystem, exemplified by the relationship between government funding and academic research. The collaboration between academia, industry, and federal agencies has been instrumental in accelerating scientific breakthroughs and fostering a culture of innovation. This synergy can be traced back to the advancements that arose during World War II, when the urgent need for medical solutions led to unprecedented cooperation among scientists, policymakers, and industry leaders. By aligning their resources and expertise, these entities launched initiatives that provided significant advancements in pharmaceuticals, medical devices, and public health strategies, ultimately proving that collective efforts could yield extraordinary outcomes in biomedical research and technology.
As we navigate contemporary health challenges, the relevance of public-private partnerships continues to be paramount. With escalating costs and complexity in modern biomedical research, these collaborations provide a crucial framework for leveraging resources effectively and driving innovation. They promote shared risk-taking among stakeholders, encouraging investment in high-reward areas of research that might otherwise remain unfunded. As seen in recent developments in vaccine research and treatment for various diseases, successful public-private partnerships harness the competing strengths of both sectors to deliver timely and impactful results. Promoting this collaborative spirit can ensure that the U.S. remains at the forefront of health innovations that not only serve national interests but also address global health challenges.
Moreover, the impact of public-private partnerships extends beyond immediate research outcomes; they also create a beneficial feedback loop that enhances the overall capacity of the U.S. innovation ecosystem. By collaborating on research projects, pharmaceutical companies gain access to cutting-edge discoveries from academic institutions, while universities benefit from the practical insights and commercial perspectives provided by industry veterans. This interchange of ideas fuels a continuous cycle of improvement and advancement in biomedicine and beyond, ultimately leading to enhanced patient care and expanded healthcare access. As these partnerships continue to evolve, they play an essential role in shaping the future of the U.S. health innovation system, ensuring its adaptability in a rapidly changing global landscape.
The Evolution of the Biomedical Innovation System
The evolution of the U.S. biomedical innovation system represents a profound journey that transforms the landscape of healthcare over the decades. Tracing its roots back to the collaborative efforts during World War II, the system has gained momentum through strategic public investments and an increasing emphasis on partnerships between academia and industry. The pre-war era was marked by rudimentary research, minimal funding, and an underdeveloped pharmaceutical industry, highlighting the necessity for a structured approach to innovation. The wartime exigencies demonstrated the potential of organized research, leading to a redefinition of what was possible in biomedicine, prompting the design of institutional frameworks that would bolster collaborative efforts in the decades that followed.
In the postwar era, numerous scientific breakthroughs emerged, underscoring the symbiotic relationship between universities and federal research bodies. Laboratories that once focused on a narrow scope of projects shifted to leverage enhanced funding for diverse research endeavors. This broader scope enabled significant advancements in drug development and therapeutic modalities, with the establishment of the NIH serving as an anchor for funding that supported pioneering research in various biomedical fields. The transformation from a disorganized pharmaceutical landscape to a structured and advanced biomedical ecosystem illustrates how sustained investment and collaboration have driven progress and innovation.
As we look to the future, the continuing evolution of the biomedical innovation system hinges on the ability to adapt to emerging scientific challenges and technological advances. Current trends, such as personalized medicine, biotechnology, and genomics, demand innovative solutions that combine traditional expertise with new scientific approaches. Policymakers and industry leaders must work together to ensure that the foundational principles established decades ago remain relevant in supporting a dynamic research environment. This commitment to fostering collaboration and investing in innovative research will define the trajectory of the U.S. biomedical innovation system and its ability to maintain global leadership in healthcare advancements.
Challenges in Biomedical Research Funding
Despite the historical success of the U.S. health innovation system, significant challenges remain regarding biomedical research funding. The increasing pressure for cost containment and budget cuts within federal funding agencies poses risks to ongoing and future research initiatives. As seen in recent policy discussions, proposed limitations on reimbursement for indirect costs could hinder the ability of academic institutions to sustain their research operations. The consequences of such funding cuts would ripple through the innovation ecosystem, potentially stalling the progress that has characterized U.S. biomedical research and undermining the public-private partnerships essential for catalyzing breakthroughs.
Furthermore, as the complexity of modern scientific challenges escalates, the need for stable funding and resource allocation becomes even more urgent. Contemporary research demands substantial investments to explore innovative therapies, especially in emerging fields like genetic engineering and personalized medicine. Researchers depend on federal funding not only to cover direct project costs but also to support overhead and training opportunities for a new generation of scientists. Fostering an environment conducive to long-term commitments in research funding is critical to ensuring a sustainable and adaptive innovation system capable of addressing both current and future health issues.
In addition to budgetary constraints, the discussions surrounding federal funding for biomedical research often raise questions about prioritization and allocation within the health innovation ecosystem. As public health crises emerge, the challenge remains to effectively balance funding across various areas of research to ensure comprehensive support. While infectious disease research, such as vaccine development, has garnered attention recently, it is essential not to overlook other critical areas that require sustained investment. The historical context of successful partnerships illustrates the risks associated with narrowing funding focus, as a holistic approach to biomedical research is fundamental for fostering a diverse landscape of innovative solutions that respond to a wide array of health challenges.
Ensuring that federal funding for biomedical research remains robust, comprehensive, and inclusive of all areas will be key to advancing the U.S. health innovation system and maintaining its global leadership. Continued dialogue among legislators, researchers, and industry stakeholders will be crucial in navigating these pressing challenges and ensuring the sustainability of an innovation ecosystem that has been the cornerstone of American biomedical advances for decades.
Frequently Asked Questions
What role does the U.S. health innovation system play in biomedical research?
The U.S. health innovation system is integral to advancing biomedical research by fostering collaborations between universities, government agencies, and the private sector. This collaborative ecosystem facilitates groundbreaking discoveries and the development of new therapies, as evidenced by historical successes like the mass production of penicillin during World War II.
How have public-private partnerships influenced the U.S. health innovation system?
Public-private partnerships are a cornerstone of the U.S. health innovation system, driving research and development across biomedical fields. These partnerships leverage federal funding and academic resources to propel innovations that enhance public health, as seen in the development of new drugs and treatments.
What is the impact of federal funding on the U.S. health innovation system?
Federal funding significantly impacts the U.S. health innovation system by providing essential resources for academic research and development. This support enables universities and research institutions to pursue scientific breakthroughs that lead to advancements in medicine and health technologies.
Can you explain the history of penicillin within the U.S. health innovation system?
The history of penicillin is a prime example of the U.S. health innovation system in action. During World War II, government-supported research facilitated the mass production of penicillin, transforming it into a lifesaving antibiotic and demonstrating the critical role of federal support in biomedical advancements.
What are some significant scientific breakthroughs in the U.S. health innovation system?
The U.S. health innovation system has produced many significant scientific breakthroughs, including antibiotics like penicillin, advanced cancer treatments, and innovative medical technologies. These breakthroughs stem from the collaboration between federal funding, research institutions, and the private sector.
How has the U.S. health innovation system evolved since World War II?
Since World War II, the U.S. health innovation system has evolved through an increased emphasis on public-private partnerships and the integration of rigorous scientific methods in research. This evolution has led to enhanced capabilities in biomedical research and a stronger pharmaceutical industry.
What are the challenges currently facing the U.S. health innovation system?
Current challenges facing the U.S. health innovation system include potential cuts in federal funding for medical research and the need to adapt funding structures for indirect costs. These challenges impact the capacity for ongoing biomedical research and the sustainability of public-private collaborations.
| Key Points |
|---|
| The U.S. health innovation system is a global benchmark shaped significantly during World War II. |
| Federal government involvement initiated the partnership between public and private sectors that drove major advancements in biomedical research. |
| Major breakthroughs, like the mass production of penicillin, emerged as a response to urgent wartime needs, showcasing the effectiveness of collaborative R&D. |
| Today’s biomedical innovation is built on a foundation laid by wartime research, infrastructure, and frameworks created during the 1940s. |
| The partnership between universities, the NIH, and the life sciences industry continues to thrive and is crucial for future advancements. |
| Current discussions around federal funding highlight the need to maintain support for this cornerstone of U.S. health innovation. |
Summary
The U.S. health innovation system stands as a testament to the nation’s pioneering spirit and collaborative efforts initiated during World War II. This unique partnership between the federal government, academia, and the private sector has revolutionized biomedical research, leading to significant breakthroughs, including the development of life-saving antibiotics. As we look to the future, it is imperative to sustain this model, ensuring that the U.S. remains at the forefront of global health innovations that benefit both national and global public health.